How to Create a Study
The Create Study interface lets you create a new Q Methodology study and set up its properties. You can also import a study to analyze the data on Q Method Software.
Follow these steps to create a new study:
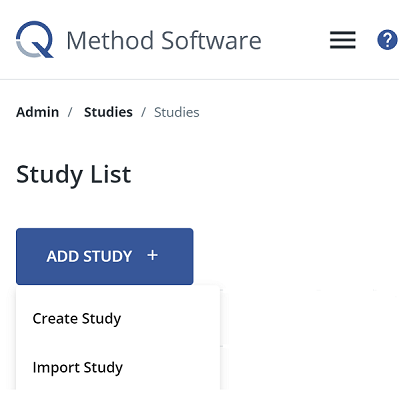
1. Open the Create Study interface
From the Study List, click Add Study +. Then, click Create Study. You will be redirected to the Create Study interface. You will progress through 3 steps to create your new study. First, you add the study details, then you enter concourse statements, and then you configure the distribution grid. Once you configure the required settings in the Study Details, you can skip through steps 2 and 3 and finish it later.
2. Add Study Details
TIP: Mandatory fields are marked with an asterisk. If you don’t fill in these fields, you will not be able to save the new study.
a. General settings:
Title: Give your study a title. The title of your study is visible to participants when they are participating in your study.
Start and end date: All studies must be configured with a start & end date and time. By default, the start date is set to the current date and time, and the end date iqs automatically set to be one month later, configure the start and end date to match your study’s requirements.
Study description: The description field is optional, and is for your internal reference only. The study description is not shown to participants.
Manage the colour scheme of your study by using one of our predefined themes: Click on the drop-down menu to view the colour schemes available for your study. Select one of the themes to change your study’s colour scheme.
- TIP: Click the yellow button on each theme to preview the colour scheme.

- TIP: Click the yellow button on each theme to preview the colour scheme.
Import theme from another study: If you have configured a theme in another study and you want to configure your new study to have that same theme, import that theme from your previous study here.
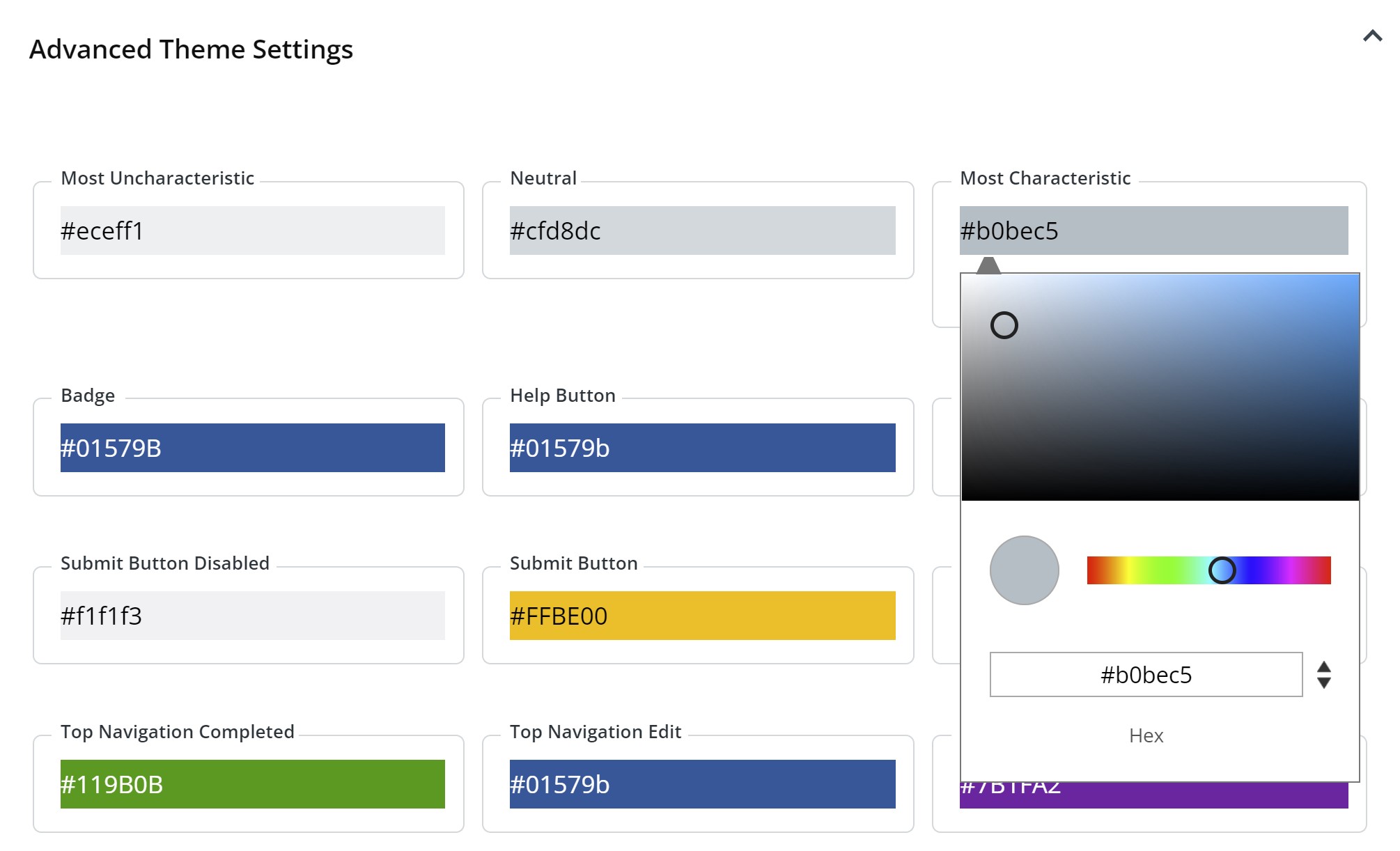
Manage the colour scheme of your study by using custom colours in these advanced settings: Configure a colour custom theme here. Choose your colour scheme based on the labels.
b. Participation code entry management:
If your study uses participation codes, you can manage the text that appears on the code-entry page of your study. If you are not using participation codes, then you do not have to configure any participation code settings.
Are you using participation codes in your study? If you are using participation codes in your study, select yes.
Allow dynamic creation of participation codes? Select yes if you want Q Method Software to create unique participant codes. Select no if you want to create custom participant codes.
Do you want to allow participants to enter the study without a participant code? If you would like to enable participants to be able to take your study without needing to give them a participation code, select yes. If you enable participants to take your study without a participation code, you can still use participation codes. When this option is enabled, you only need to provide participants with the study link, and they will be asked if they have a participation code or not.
Participant code selection text: This group of settings allows you to customise the text that will appear to your participants on the first page of the study. You can customise the labels and buttons that appear when participants specify whether they have a participation code.
- WARNING: if you selected the option to not allow participation codes in your study, these settings will not appear.
- TIP: Adding custom text to these fields is optional. If you do not add your own text, then default text settings will appear for each of these fields.
User has participation code button text: The text that appears on the button to indicate the participant has a participation code.
User doesn’t have participation code button text: the text that appears on the button to indicate the participant does not have a participation code.
Participation code entry box label: the text that appears on the interface where the participant enters their participation code.
Submit button text: the text that appears on the button that the participant clicks to submit their participation code for verification.
Cancel button text: the text that appears on the button that the participant clicks to cancel entering a participation code, which brings them back to the starting point of the study.
Error message when code is not entered: the validation error message that appears when a participant did not enter a participation code, but clicked the submit button.
Error message when code is not found: the validation error message that appears when a participant enters an invalid participation code. (eg: code not found).
c. Scale management:
Manage the labels and icons of the three scales that appear on the distribution grid to help your participants rank the statements. You can click the scale management settings to preview how the labels will look to your participants.
Display timer: If you want your study to have a timer visible to your participants, select display timer.
Display scale numbers: Scale numbers appear on the bottom of the distribution grid. If you want to display scale numbers to your participants, select display scale numbers.
Display neutral anchor: by default, the q-sort distribution will contain two anchors on either side of the distribution: one to indicate, for example, the most representative group of statements, and one to indicate the least representative group of statements. By checking the Display Neutral Anchor option, the word "Neutral" will appear exactly between the two anchors, indicating the most neutral group of statements.
Low scale label: you can change the low scale icon by selecting choose a file. You can also change the scale label by entering a custom label in the text field.
Neutral scale label: you can change the neutral scale icon by selecting choose a file. You can also change the scale label by entering a custom label in the text field.
High scale label: you can change the high scale icon by selecting choose a file. You can also change the scale label by entering a custom label in the text field.
TIP: You are not required to change the scale labels. If you do not change the scale labels, the default labels will appear to your participants.
d. Email management:
These settings allow you to configure invitation emails, reminder emails to send to participants, and the frequency of the email reminders.
Invitation email template: type or paste an invitation email in the text field. This content will appear in the invitation email that can be optionally sent to participants through Q Method Software. Invitation emails can contain placeholders to provide participant-specific information, such as their unique participation code.
Reminder emails: allows you to select if or when reminder emails should be sent to participants who have been sent an invitation email, but have yet to complete their study. You can select one of the following options:
- Do not send reminder email: disables reminder emails from being sent to participants who received an invitation email.
- One reminder, 7 days after invitation email: send a reminder once, one week after the initial invitation email was sent.
- Reminder every 7 days after invitation email: send a recurring reminder every week after the initial invitation email until the participant completes their study.
Reminder email template: this is the content that will appear in the reminder email that you can optionally send to participants. You can also send email reminders manually at any point after sending the invitation. The reminder email template will also be used if you send reminder emails manually.
Email placeholders: When adding content to your invitation or reminder email, you can add placeholders for unique variables, such as a participant’s unique participation code, to enable form mail functionality.
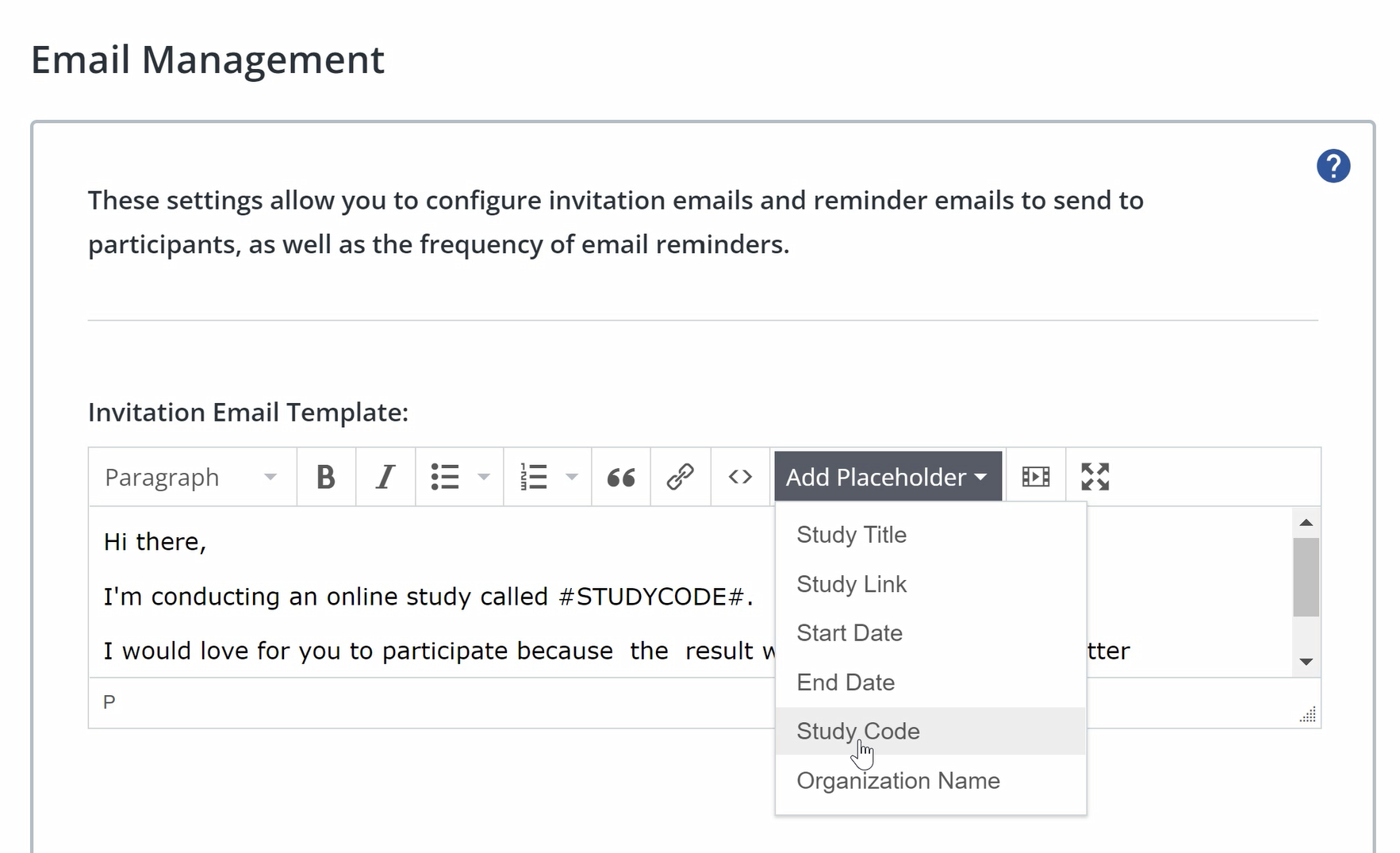
To add a placeholder, click add placeholder in the text field, and select the placeholder you want to use. The available placeholders are:
- Study Title: the title of your study will appear in the place of the placeholder.
- Study Link: a clickable link to your study, refilled with the participant’s participation code will appear in the place of the placeholder.
- Start Date: the start date of your study will appear in the place of the placeholder.
- End Date: the end date of your study will appear in the place of the placeholder.
- Study Code: the unique participation code that is assigned to the participant will appear in the place of the placeholder.
- Organisation Name: the organisation name that is configured in your profile will appear in the place of the placeholder.
e. Consent management:
Once the participants enter the study, the consent form will appear. These settings allow you to manage what content appears to the participants on the consent page.
Disable consent step: select this if you do not want to use the consent step. Participants will be brought to the first step of the study after consent if selected.
Consent text: paste or type your consent content here. The consent text must outline and disclose what your participants are consenting to.
Agree button text: the text that appears on the button for the participant to agree to the consent.
Do not agree button text: the text that appears on the button the participant to signify that they do not agree with the consent.
f. Instruction management:
This group of settings allows you to manage the content for your study’s instruction step which is displayed to participants after they have given their consent.
Instruction text: Paste or type the specific instructions for your participants to follow. Remember, you can use placeholders and link to external websites or downloadable files.
- TIP: You can add images, audio, and video on the instruction page. For a comprehensive video tutorial for participants, check out our video: How to complete a Q-Sort.
Next step button text: the text that appears on the button for the participant to go to the next step of your study.
g. Stimulus management:
This group of settings allows you to manage the content for your study’s stimulus step (which is an optional step). Prior to the presort and q-sort steps of the study, you can present stimuli such as text, video, or images to your participants. Use the field below to add any content to the stimulus page of your study.
Restrict stimulus media playback: select this option if you want participants to be able to view the video only once. Leave it unchecked if you want them to be able to replay the video as many times as they wish.
Stimulus: Paste or type the content for the stimulus that you would like to display to your participants. You can format the text, insert hyperlinks, and use placeholders. To insert a video, click the insert/edit media icon. You can paste a URL of a video or you can embed the video. Click Ok to insert the media.
h. Presort management:
These settings allow you to manage the presort step where participants initially sort statements according to the scales that you have configured. You may choose to bypass the presort step altogether.
Bypass presort step: If you want to skip the presort step, click bypass presort step. If you want to include the presort step, you can configure the test of the presort management settings.
Randomise pre-sort statements: if you check this box, then all of the statements will be randomly ordered for participants to sort.
Require participants to pre-sort all statements before proceeding to q-sort: if you check this box, then participants cannot proceed to the next step unless all the statements are sorted.
Presort text: content that appears above the cards to be presorted during the presort step. This can include short instructions or hints to help your participants with their sorting.
Next step button text: the text that appears on the bottom of the page for participants to go to the next step of your study.
Presort completed message: the text that appears one the pre-sorting is complete.
TIP: adding custom text to these fields is optional. If you do not add your own text, default text will appear for each of these fields.
i. Final sort instructions:
These settings allow you to provide custom instructions for the final sort of your study.
Instruction text: Paste or type the instructions that will appear to participants on the final sort step of the study.
Next step button text: the text that appears on the button for participants to go to the next step of your study.
j. Q-sort management:
On the final qsort step, participants place their statements on the qsort distribution grid. There are various text settings you can configure to your specific study. Adding custom text to the fields below is optional. If you do not add your own text, default text will appear for each of these fields.
Q-sort help text: you can optionally add detailed help content for your participants to access when they are performing their q-sort task. This content is displayed when the participant clicks the help icon in the q-sort step. If you leave this blank, the default help content will appear.
Help icon tooltip text: the text that appears when the participant hovers over the help icon. You can customise the text to be a different language or to be more pertinent to your study.
Reset q-sort tooltip text: the text that appears when the participant hovers over the reset q-sort icon.
Submit q-sort disabled tooltip text: the text that appears to participants when they hover over the submission icon to submit, but the q-sort task has not been completed yet.
Submit q-sort confirmation panel text: the text that appears in the confirmation panel when the user has clicked the submit icon, which asks the user if they really want to submit the study.
Submit q-sort confirmation panel yes button text: the text for the button in the confirmation panel to indicate that the participant wishes to submit their completed study.
Submit q-sort confirmation panel no button text: the text for the button in the confirmation panel to indicate that the participant does not want to submit their completed study.
Allow participants to expand card piles: Participants can expand card piles to view multiple statements at once while sorting their statements. To enable this setting, click allow participants to expand card piles.
Expand pile tooltip text: the text that appears when a participant hovers over the expand pile tooltip.
k. Completion management:
After participants submit their q-sort, they are presented with a final page to indicate the study’s completion. These settings control what content is displayed on the final page.
Completion text: Paste or type the text that will appear on the final page of the study. You can format the text, create hyperlinks, and use placeholders.
Redirect to URL: if you want to redirect participants to an external webpage after completing the study, enter the URL in the field below. You can include placeholders in the URL to pass data with the URL such as the participation code or the instance ID. Click the Placeholders button once you enter the URL, then select the item that you wish to insert into the URL. You can change the default parameter names that are inserted by clicking into the text box and manually changing the parameter names.
l. Navigation label management:
At the top of the study page, there are navigation labels for each step of the study. You can change any label name by editing the text field below it. Keep in mind that label names are limited to 25 characters.
Code-entry navigation text
Consent navigation text
Instructions navigation text
Stimulus navigation text
Pre-sort navigation text
Final sort instructions navigation text
Final-sort navigation text
Finish navigation text
Survey navigation text
m. Survey management:
- Submit survey error: If a participant encounters an error while submitting a survey response, an error message will pop up. You can modify the error message by changing the text below
3. Next Step
Once you have completed the Study Details, you can click save and add concourse statements. You will be redirected to the second step of the study creation process.

4. Enter Concourse Statements
In this step, you will add the statements that make up the q-sort concourse.
- Click Add Statements. If you have text-based statements, you can paste or type your statements in the text section. Make sure that you only have one statement per line. If you have image-based items, click the image option, and then upload images to populate the concourse statements.
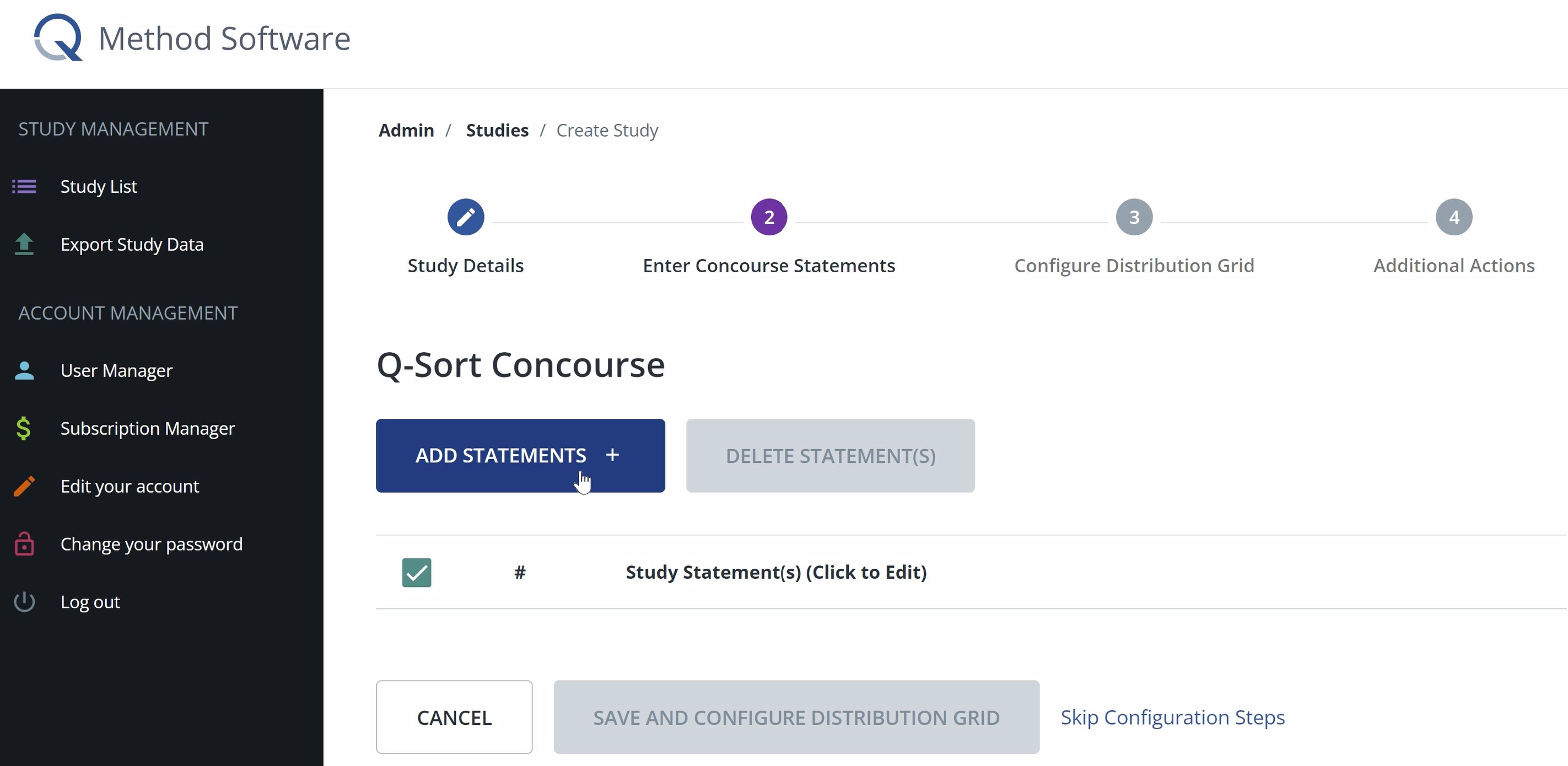

- Once you have added all your statements, click submit. The statements should appear in a configurable list. You can drag and drop the statements to modify the order in which they appear to participants.
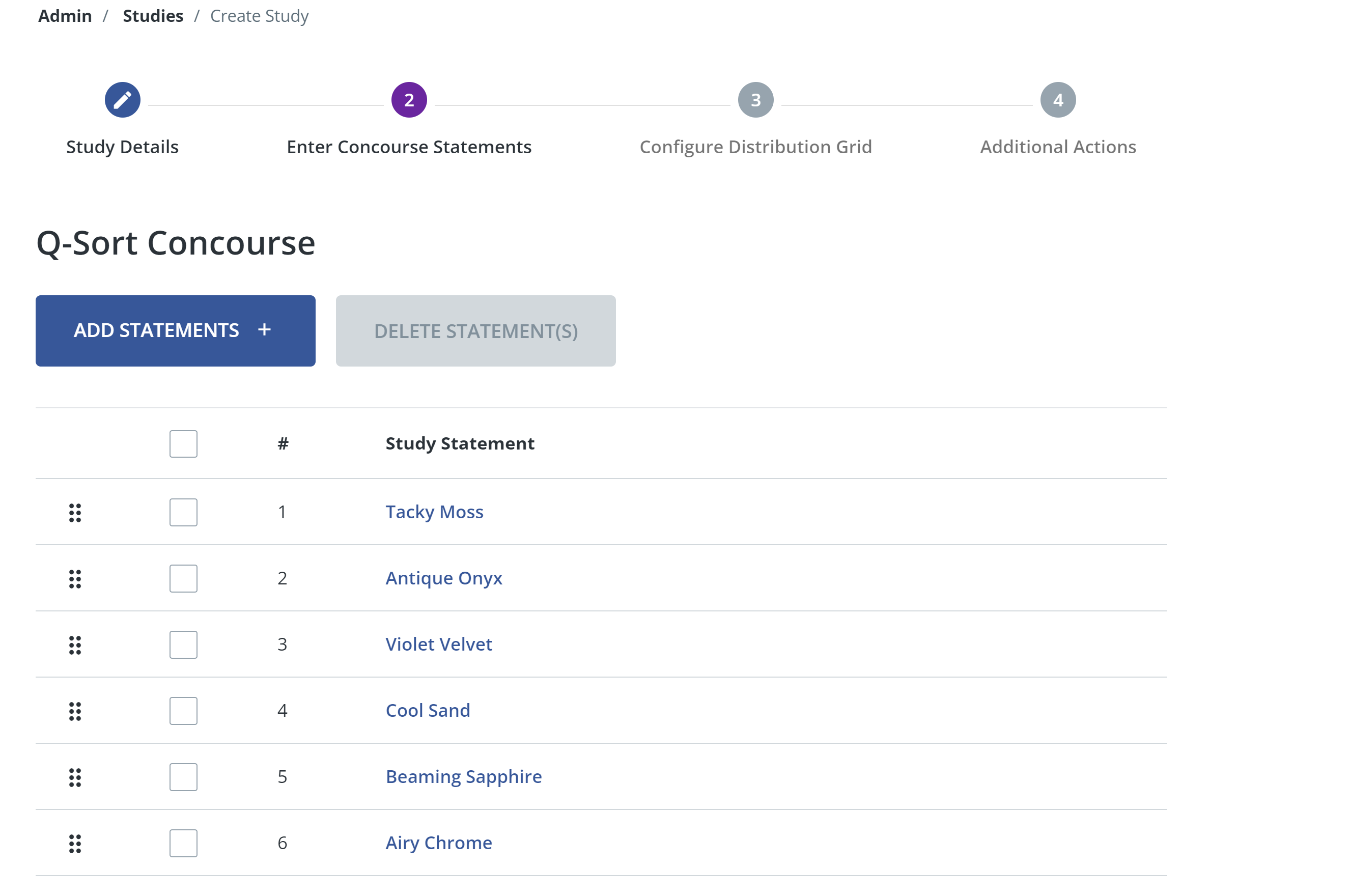
- TIP: you can add/edit your concourse after the initial study setup. However, you can only change your concourse if you have not yet started receiving q-sorts. If participants have begun completing and submitting q-sorts, the concourse will be locked and you will not be able to make changes anymore.
- To progress to the next step of the study creation process, click save and configure distribution grid.
5. Configure distribution:
In this step, you will configure the distribution grid based on the amount of statements entered in the previous step.
- First, choose whether the grid orientation is bottom-based or top-based. If you choose bottom-based, the distribution grid base and labels will appear along the bottom of the grid. If you choose top-based, the grid base and labels will appear along the top of the grid.
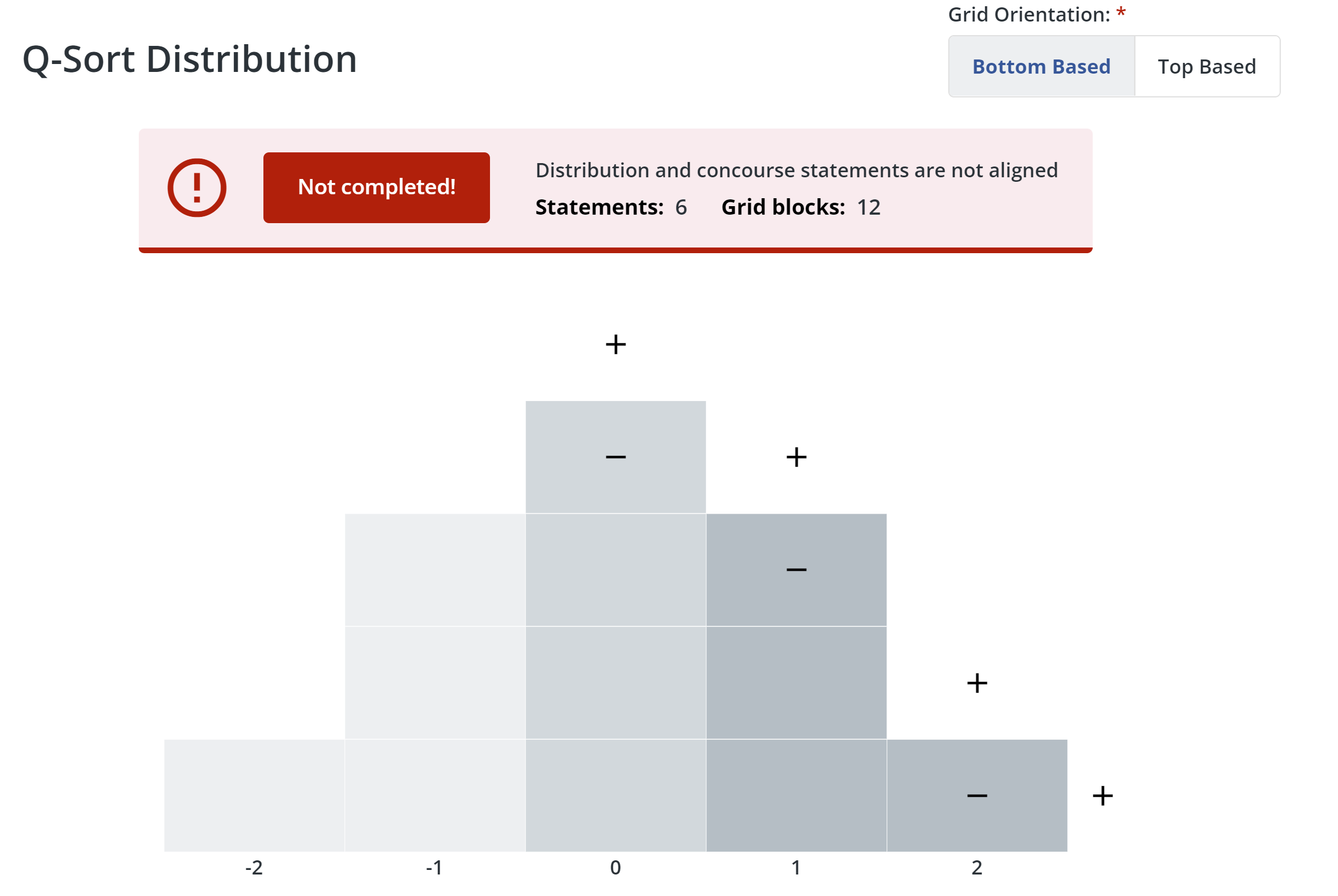
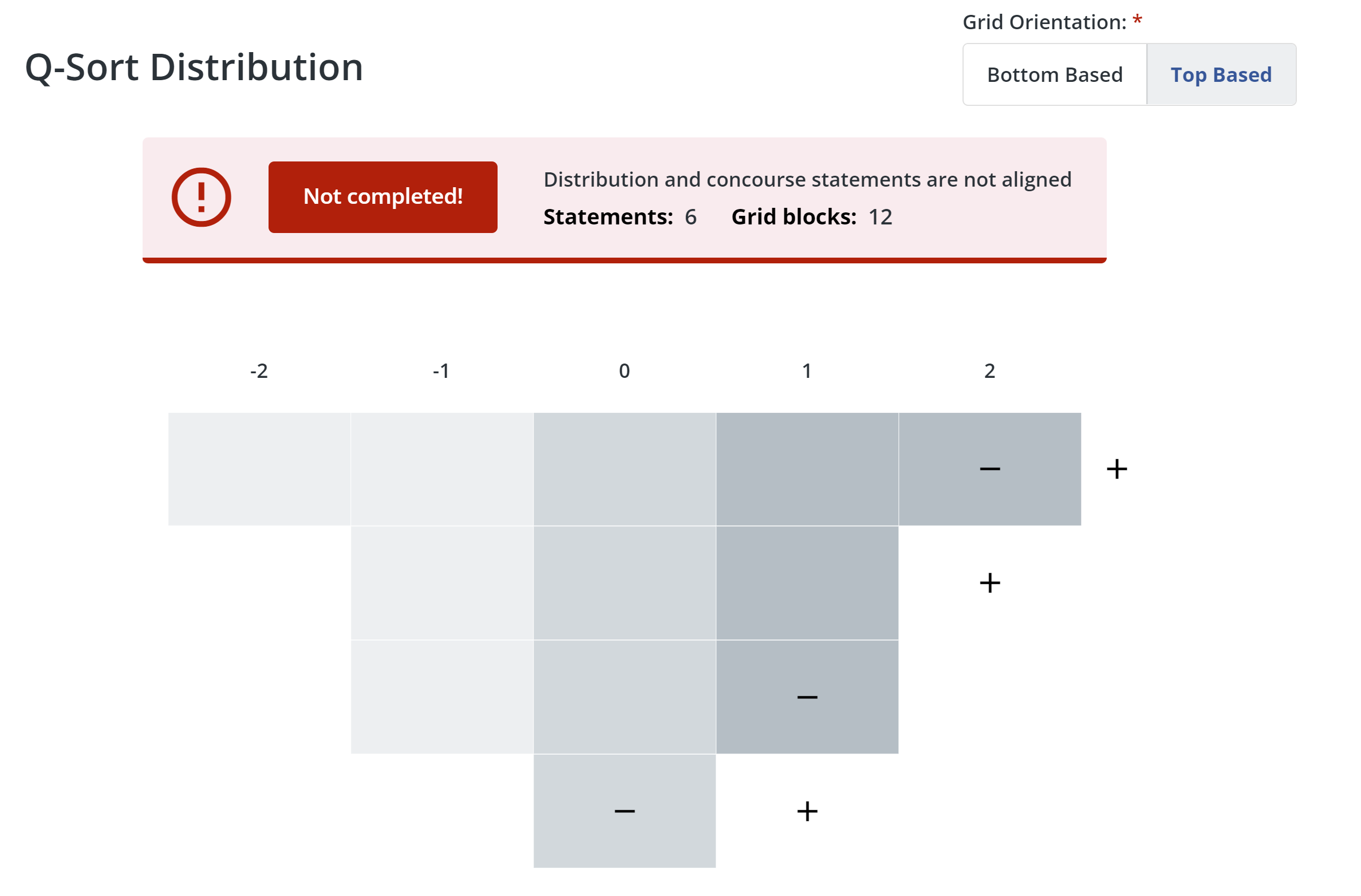
- Q Method Software lets you know if you need to add or remove blocks on the distribution grid to match the number of statements you have. Click + or - to add or remove columns and blocks. When the number of blocks in the grid matches the number of statements in your study, the message box will turn green.
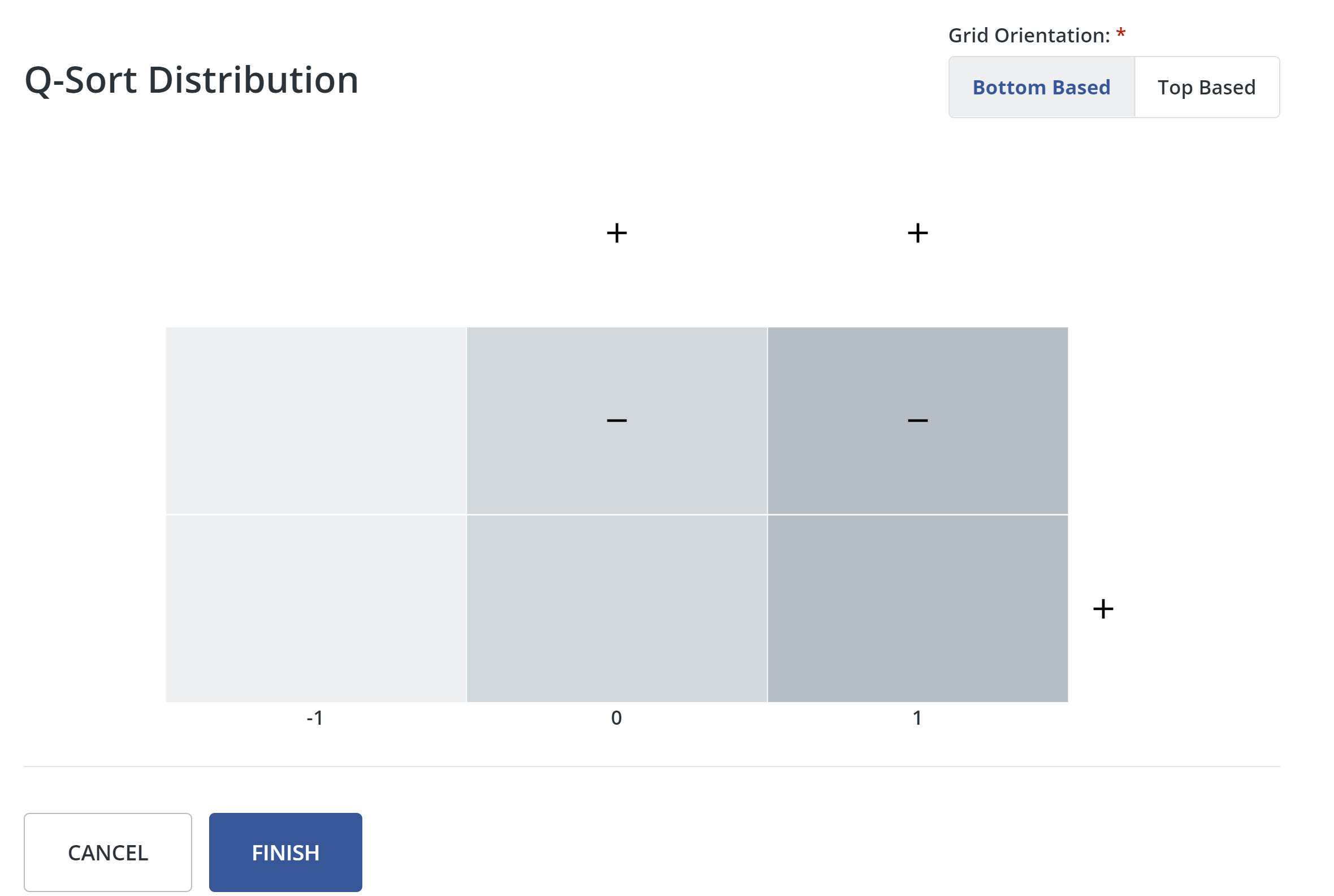
- When you are finished configuring the distribution grid, click save and finish.
6. Submit
Next, you will be redirected to a fourth page in the study creation process. You can view a summary of your study’s settings and see which settings still need to be configured. You may also choose to add a survey, manage your participants, or be redirected to your study’s dashboard.
Import Study
To import a study, follow the instructions below:
From the study list, click Add Study. Select Import Study from the drop-down menu.
General study details: Give your study a title and a description. You can view and manage other study settings after you import your data.
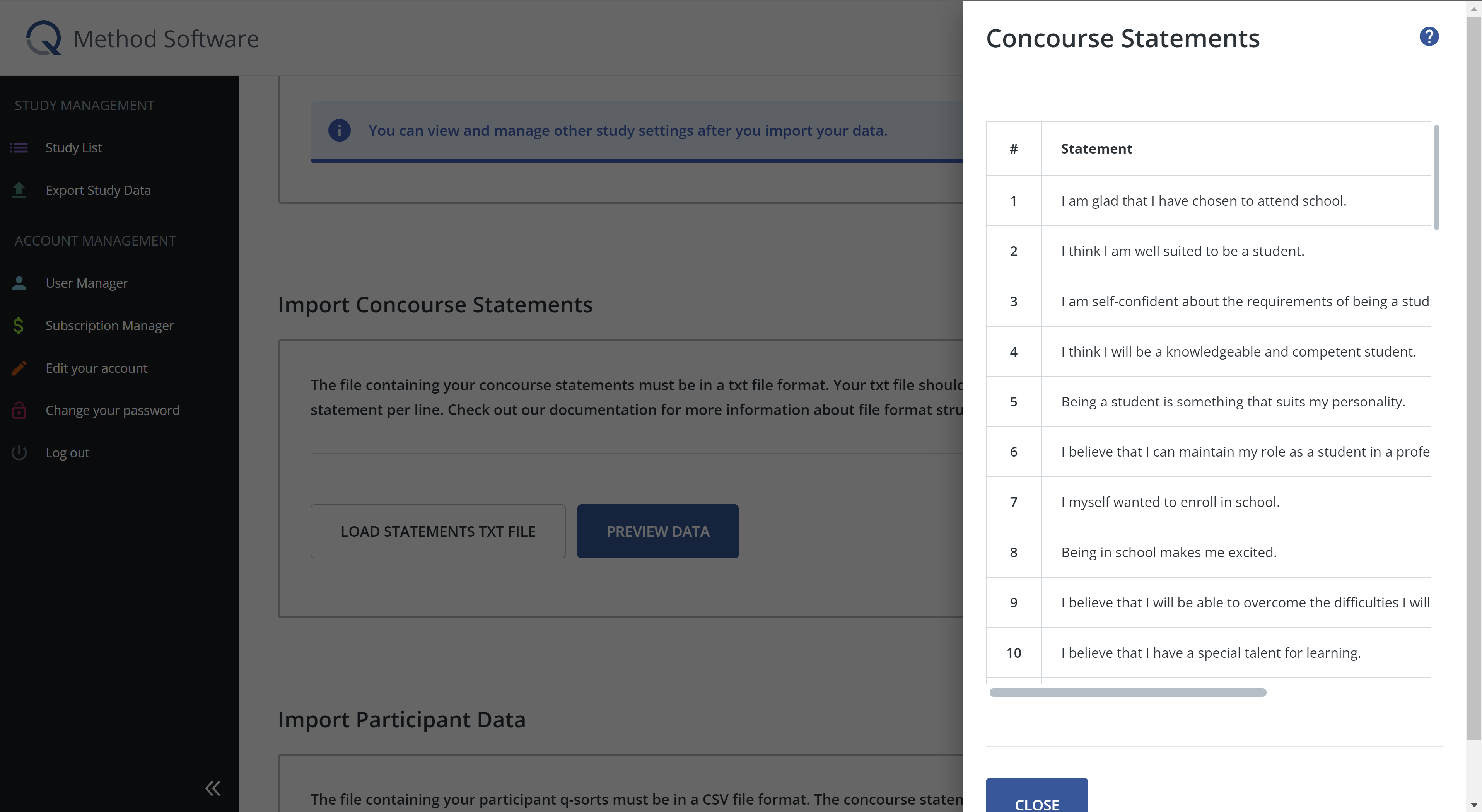
Next, you will import your concourse statements. The file containing your concourse statements must be in a txt file format. Your txt file should have one concourse per line (see below for an example). To upload the statements, click load statements txt file and select the txt file from your device. Once the file is loaded, click preview data to confirm that the statements appear correctly. If your file is not uploading correctly, it is most likely a file formatting error.

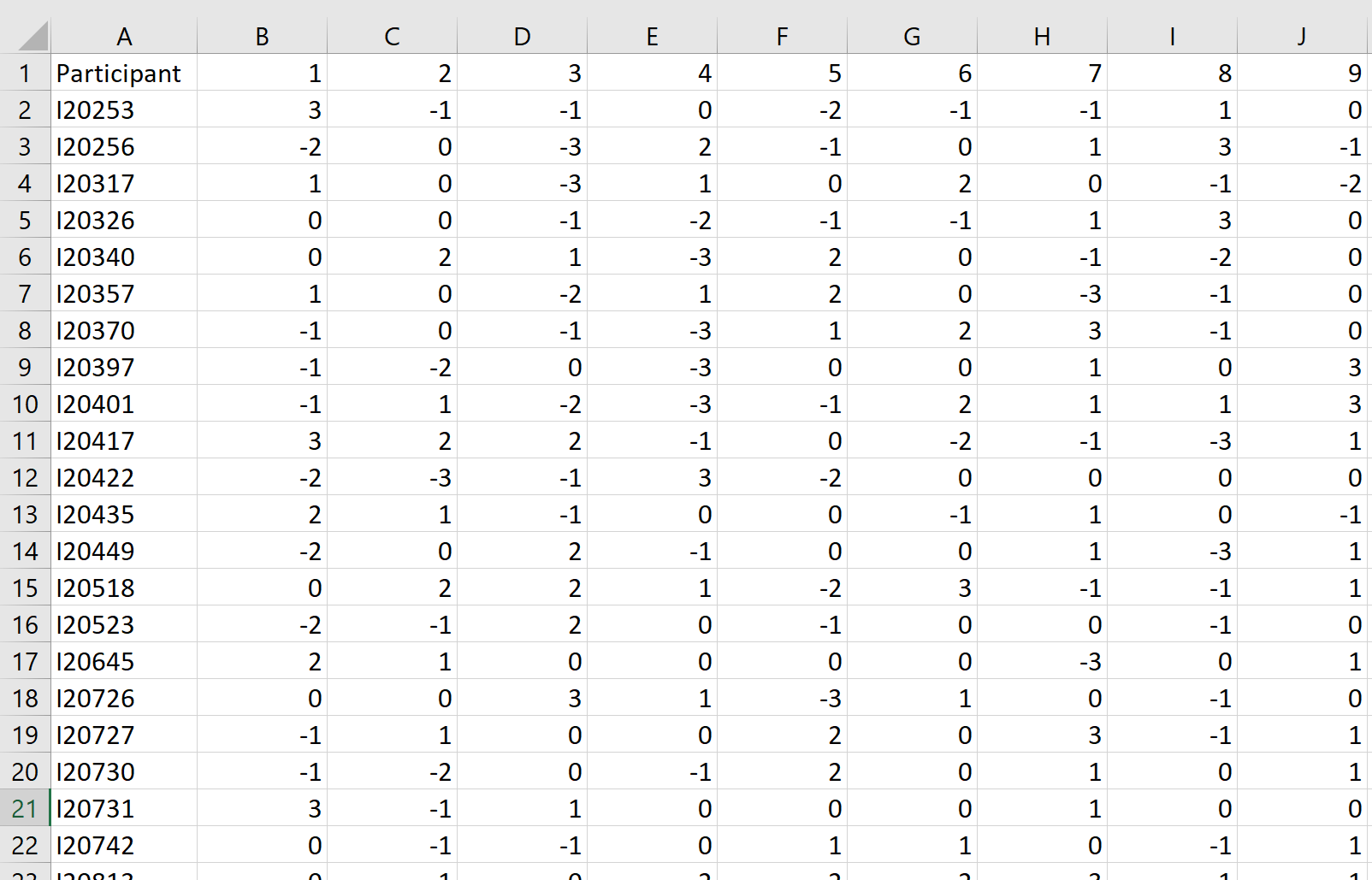
Next, you will import your participant data. The file containing your participant q-sorts must be in a CSV file format. The concourse statement numbers should be contained within the first row, and the participant data must be on one line (see below for an example). Once the file is loaded, click preview data to confirm that the data is loaded correctly.
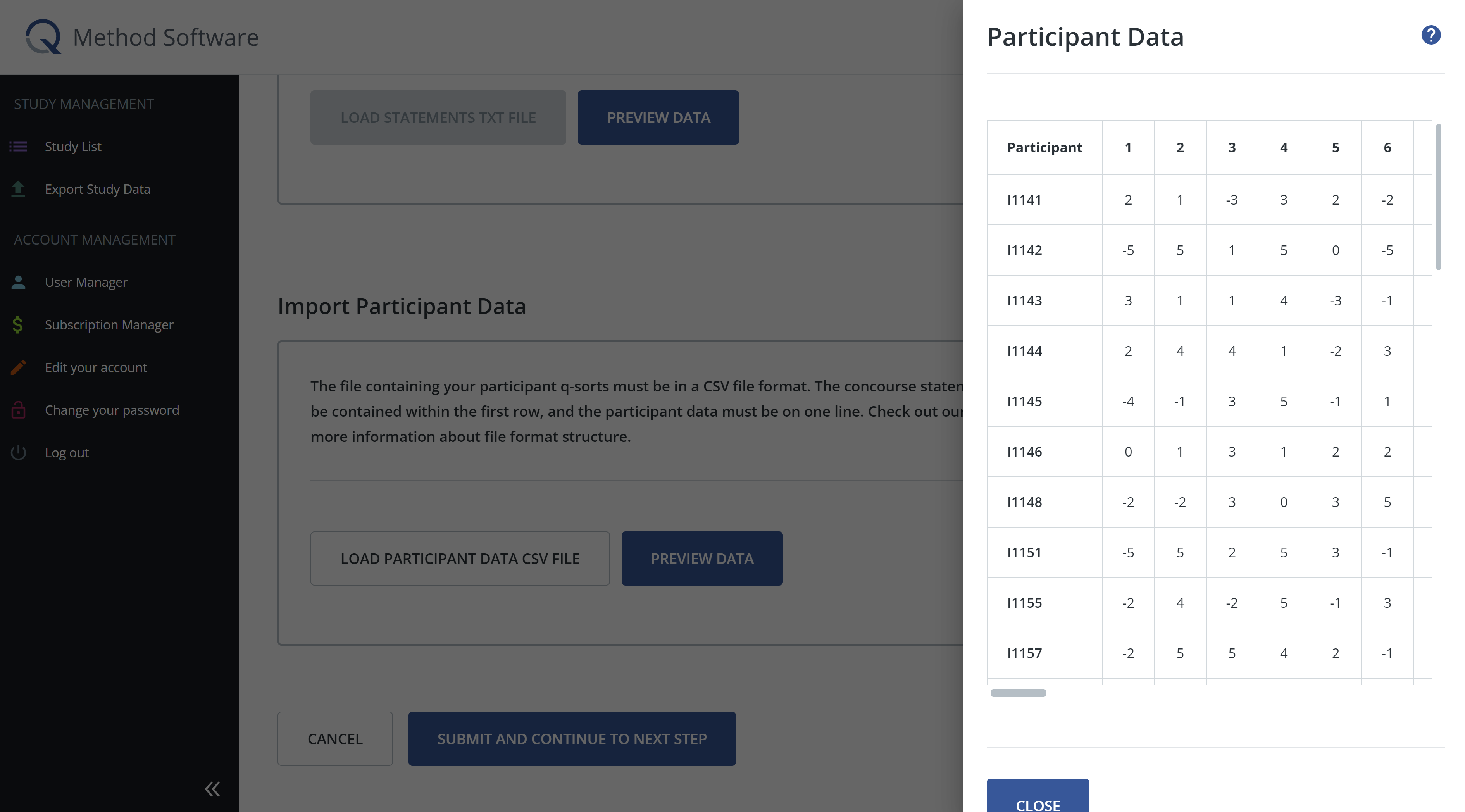
Once you have added concourse statements and/or participant data, click submit and continue to the next step.
You will be redirected to a completion summary page that outlines the details of your study.